-
Prestige Biopharma drug gets positive EMA opinion on Orphan Designation to treat pancreatic cancer
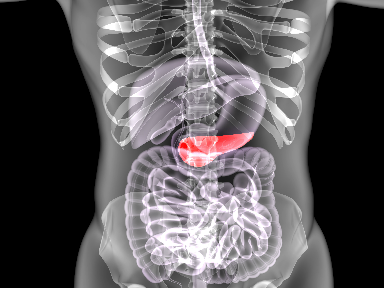
October 19, 2020Prestige BioPharma announced that the European Medicines Agency (EMA) Orphan Drug Commission (COMP) has granted a positive opinion for an Orphan Drug Designation (ODD) status to its first-in-class anti-PAUF monoclonal antibody, PBP1510, for the ...
 ALL
ALL Pharma in China
Pharma in China Pharma Experts
Pharma Experts Market News
Market News Products Guide
Products Guide Brand Story
Brand Story






 Pharma Sources Insight January 2025
Pharma Sources Insight January 2025








