-
InnoCare Announces First Patient Dosed in the Phase III Registrational Trial of BCL2 Inhibitor ICP-248 in Combination with Orelabrutinib as First-Line Therapy for CLL/SLL Patients in China
PharmaSources
March 28, 2025
InnoCare announced the first patient dosed in the Phase III trial of ICP-248 combined with orelabrutinib for CLL/SLL. It's a potential treatment, with other trials advancing.
-
InnoCare Releases 2024 Results and Business Highlights
PharmaSources
March 27, 2025
InnoCare announced 2024 annual results, including financials, R&D progress in oncology and autoimmune diseases, and globalization efforts.
-
InnoCare Announces First Patient Dosed in the Phase III Registrational Trial of ICP-488 for the Treatment of Psoriasis in China
PharmaSources
March 20, 2025
InnoCare announced the first patient dosing in the Phase III trial of ICP - 488 for psoriasis. It showed good results in Phase II, aiming to meet unmet needs.
-
Data of InnoCare's ICP-488 for the Treatment of Psoriasis Presented at Late Breaking Oral Presentation of 2025 AAD
PharmaSources
March 11, 2025
InnoCare Pharma; TYK2 inhibitor ICP - 488; psoriasis; 2025 AAD Annual Meeting; clinical data
-
InnoCare Announces the Approval of Registrational Phase III Study of BCL2 Inhibitor ICP-248 in Combination with Orelabrutinib as First-Line Therapy for Treatment of CLL/SLL Patients in China
PharmaSources
February 18, 2025
InnoCare Pharma announced on Feb. 17, 2025 that CDE approved the Phase III trial of ICP - 248 combined with orelabrutinib for first - line CLL/SLL treatment in China.
-
InnoCare Releases 2024 Interim Results and Business Highlights
August 21, 2024
InnoCare Pharma announced the 2024 interim results and corporate update as of 30 June 2024.
-
InnoCare Announces First Subject Dosed in Clinical Trial of TYK2 Inhibitor ICP-332 in the U.S.
July 24, 2024
Beijing, July 24, 2024 – InnoCare Pharma (HKEX: 09969; SSE: 688428), a leading biopharmaceutical company focusing on the treatment of cancer and autoimmune diseases
-
Data of InnoCare’s Robust Pipelines Presented at the European Hematology Association (EHA) 2024 Hybrid Congress
PharmaSources
June 14, 2024
Data of InnoCare’s (HKEX: 09969; SSE: 688428) robust pipelines were presented at the European Hematology Association (EHA) 2024 Hybrid Congress.
-
InnoCare Announces Clearance of Clinical Trial of TYK2 Inhibitor ICP-332 by U.S. FDA
PharmaSources
June 11, 2024
InnoCare Pharma (HKEX: 09969; SSE: 688428), a leading biopharmaceutical company focusing on the treatment of cancer and autoimmune diseases, announced today that the U.S. Food and Drug Administration (FDA) has cleared the Investigational New Drug (IND) ap
-
InnoCare Announces First Patient Dosed in Clinical Trial of BCL2 Inhibitor ICP-248 in Combination with Orelabrutinib as First-Line Therapy for Chronic Lymphocytic Leukemia /Small Lymphocytic Lymphoma
PharmaSources
May 21, 2024
The first patient has been dosed in a clinical trial of the Company's BCL2 inhibitor, ICP-248, in combination with orelabrutinib as a first-line therapy for chronic lymphocytic leukemia /small lymphocytic lymphoma (CLL/SLL) in China.

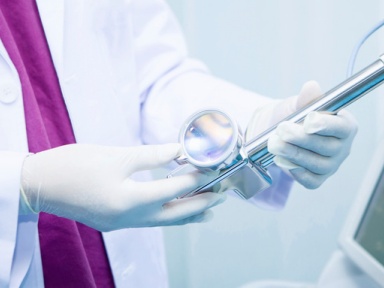

 InnoCare Pharma announced the 2024 interim results and corporate update as of 30 June 2024.
InnoCare Pharma announced the 2024 interim results and corporate update as of 30 June 2024. Beijing, July 24, 2024 – InnoCare Pharma (HKEX: 09969; SSE: 688428), a leading biopharmaceutical company focusing on the treatment of cancer and autoimmune diseases
Beijing, July 24, 2024 – InnoCare Pharma (HKEX: 09969; SSE: 688428), a leading biopharmaceutical company focusing on the treatment of cancer and autoimmune diseases


 ALL
ALL Pharma in China
Pharma in China Pharma Experts
Pharma Experts Market News
Market News Products Guide
Products Guide Brand Story
Brand Story






 Pharma Sources Insight January 2025
Pharma Sources Insight January 2025








