PhamaSources.comDecember 10, 2021
Tag: LianBio , infigratinib
· Initiated and completed dosing in pharmacokinetic (PK) study of mavacamten in healthy Chinese volunteers
· Initiated Phase 2a clinical trial of infigratinib in Chinese patients with gastric cancer and other advanced solid tumors
· Strengthened leadership team with appointment of scientific and commercial executives
· Completed initial public offering for gross proceeds of approximately $334.5 million, providing cash runway through 2023
December 9, 2021 ,LianBio (Nasdaq: LIAN), a biotechnology company dedicated to bringing innovative medicines to patients in China and other major Asian markets, today provided a corporate update and reported financial results for the third quarter ended September 30, 2021.
“Over the past several months, the LianBio team has achieved multiple meaningful milestones, including initiating our first clinical trial, completing dosing in our first PK study and successfully completing our initial public offering,” said Yizhe Wang, Ph.D., Chief Executive Officer of LianBio. “In the year ahead, we intend to continue to advance our pipeline of innovative medicines and expect to initiate four pivotal studies to support regulatory approval in our territories. I’m confident we have the leadership and expertise at hand, as well as the capital resources necessary to deliver on our commitment to bring transformative medicines to patients across Asia.”
Initiated and completed enrollment and dosing in pharmacokinetic study of mavacamten
· In November, LianBio initiated and completed enrollment and dosing in a pharmacokinetic (PK) study of mavacamten in healthy Chinese volunteers.
Initiated Phase 2a clinical trial of infigratinib in gastric cancer and other advanced solid tumors
· In August, LianBio announced that the first patient was dosed in a Phase 2a clinical trial of infigratinib in locally advanced or metastatic gastric cancer or gastroesophageal junction adenocarcinoma with fibroblast growth factor receptor-2 (FGFR2) gene amplification and other advanced solid tumors with FGFR genomic alterations.
Appointed two independent directors to LianBio Board of Directors
· In October, LianBio appointed Jesse Wu to the Company’s Board of Directors. Mr. Wu is the former Chairman of Johnson & Johnson China.
· In October, LianBio appointed Susan Silbermann to the Company’s Board of Directors. Ms. Silbermann is the former Global President, Emerging Markets at Pfizer.
Strengthened LianBio leadership team with key China-based hires
· In October, LianBio appointed Michael Humphries, MBBS as Chief Scientific Advisor to guide the Company’s research and development (R&D) strategy, advance the Company’s pipeline, and lead assessment of new in-licensing opportunities.
· In August, LianBio appointed Pascal Qian as China General Manager to build out the Company’s operations and commercial infrastructure.
Completed Initial Public Offering
· In November, LianBio completed an initial public offering (IPO) of its ordinary shares through the sale and issuance of 20,312,500 American Depositary Shares (ADSs) at a public offering price of $16.00 per ADS. Following the close of the IPO, pursuant to the partial exercise of their option to purchase additional ADSs, the underwriters purchased an additional 593,616 ADSs at the IPO price of $16.00 per ADS.
· LianBio received gross proceeds of $334.5 million in connection with the IPO and subsequent exercise of the underwriters’ option and aggregate net proceeds of $311.1 million after deducting underwriting discounts and commissions.
Mavacamten
Bristol Myers Squibb (BMS)-partnered cardiac myosin inhibitor in development for the treatment of hypertrophic cardiomyopathy and certain forms of heart failure
· LianBio expects to initiate the Phase 3 EXPLORER-CN trial of mavacamten in Chinese patients with obstructive hypertrophic cardiomyopathy in the first quarter of 2022 to support regulatory approval in China.
· LianBio’s partner BMS has announced a Prescription Drug User Fee Act (PDUFA) target action date of April 28, 2022 for the Company’s New Drug Application to the U.S. Food and Drug Administration (FDA) for mavacamten for the treatment of patients with symptomatic obstructive hypertrophic cardiomyopathy (oHCM).
TP-03
Tarsus Pharmaceuticals-partnered GABA-Cl channel blocker in development for the treatment of Demodex blepharitis (DB) and meibomian gland disease
· LianBio expects to initiate a Phase 3 trial of TP-03 in Chinese patients with DB in the second half of 2022 to support regulatory approval in China.
· LianBio’s partner Tarsus has announced that the Company expects to report topline data in the first quarter of 2022 from the ongoing Phase 3 Saturn-2 trial of TP-03 in DB patients.
NBTXR3
Nanobiotix-partnered radioenhancer in development for multiple solid tumor indications
· LianBio expects to begin dosing Chinese patients in Nanobiotix’s planned global pivotal Phase 3 trial of NBTXR3 for the treatment of locally advanced head and neck squamous cell carcinoma in elderly patients ineligible for cisplatin in the second half of 2022.
LYR-210
Lyra Therapeutics-partnered anti-inflammatory implantable drug matrix in development for the treatment of surgically-naïve, medically refractory chronic rhinosinusitis
· LianBio expects to begin dosing Chinese patients in Lyra’s planned global pivotal Phase 3 trial of LYR-210 for the treatment of surgically naïve chronic rhinosinusitis in the second half of 2022.
Research & Development Expenses
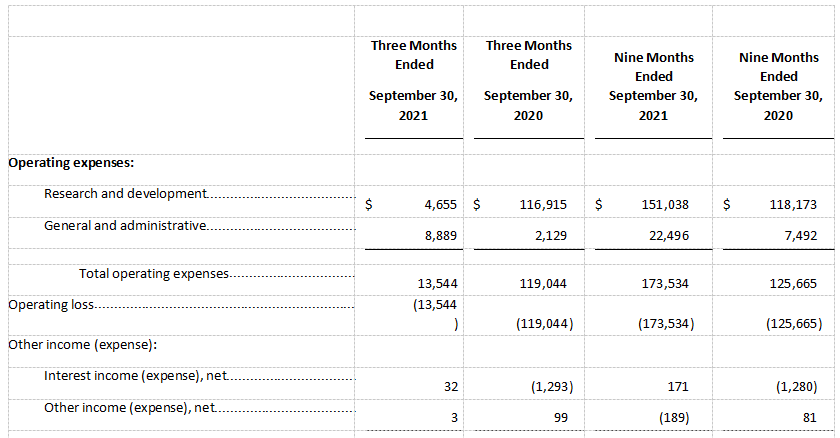
R&D expenses were $4.7 million for the three months ended September 30, 2021, as compared to $116.9 million for the three months ended September 30, 2020. For the three months ended September 30, 2021, research and development cost was primarily attributable to $1.9 million in personnel-related expenses and $2.1 million in professional fees for development activities to support clinical trials.
General & Administrative Expenses
G&A expenses were $8.9 million for the three months ended September 30, 2021, as compared to $2.1 million for the three months ended September 30, 2020. The increase was primarily attributable to increases in payroll and personnel-related expenses (including share-based compensation expense) for increased employee headcount and increases in legal service costs, consulting costs and accounting services.
Net Loss
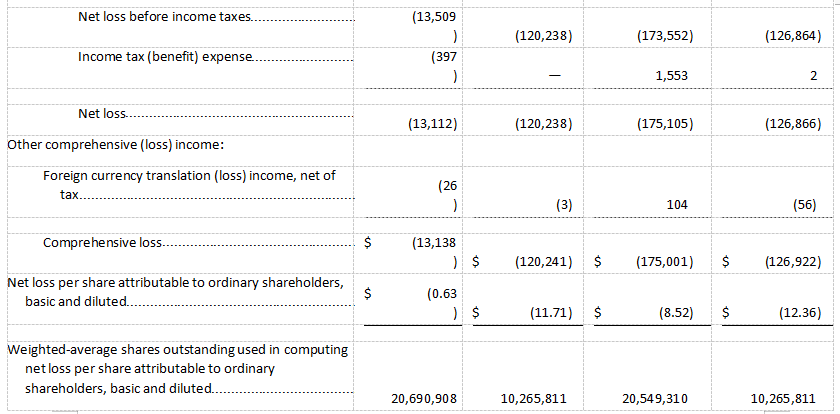
Net loss was $13.1 million for the three months ended September 30, 2021, or a net loss per share of $0.63, as compared to a net loss of $120.2 million for the three months ended September 30, 2020, or a net loss per share of $11.71.
Cash and Cash Equivalents
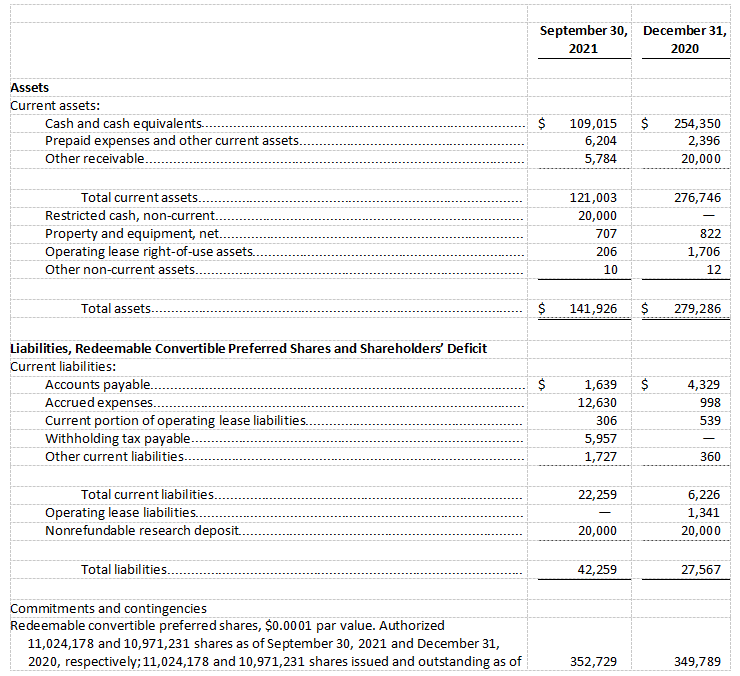
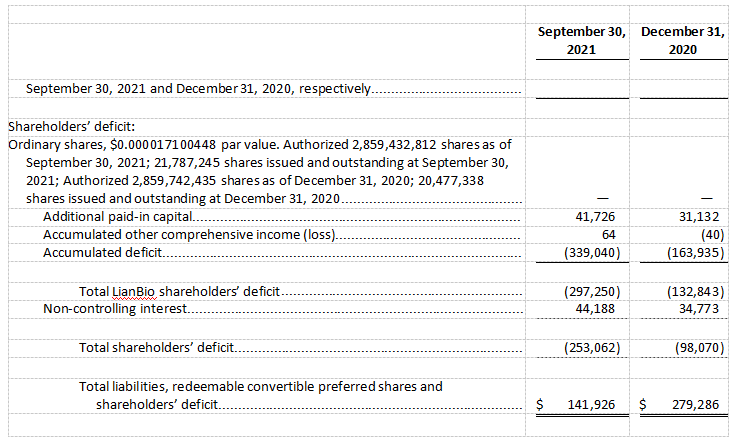
Cash and cash equivalents were $109.0 million as of September 30, 2021, which excludes the net proceeds of $311.1 million from the Company’s initial public offering, as compared to $254.4 million as of December 31, 2020. The Company expects its current cash and cash equivalents, inclusive of the IPO net proceeds subsequently received in November 2021, will be sufficient to fund its operating expenses and capital expenditure requirements through 2023.
About LianBio
LianBio is a cross-border biotechnology company on a mission to bring transformative medicines to historically underserved patients in China and other Asian markets. Through partnerships with highly innovative biopharmaceutical companies around the world, LianBio is advancing a diversified portfolio of clinically validated product candidates with the potential to drive new standards of care across cardiovascular, oncology, ophthalmology, inflammatory disease and respiratory indications. LianBio is establishing an international infrastructure to position the company as a partner of choice with a platform to provide access to China and other Asian markets.
Consolidated Statements of Operations and Comprehensive Loss
(In thousands, except share and per share data)
(Unaudited)

Consolidated Balance Sheets
(In thousands, except share and per share amounts)
(Unaudited)


Contact Us
Tel: (+86) 400 610 1188
WhatsApp/Telegram/Wechat: +86 13621645194
Follow Us:




 Pharma Sources Insight January 2025
Pharma Sources Insight January 2025


