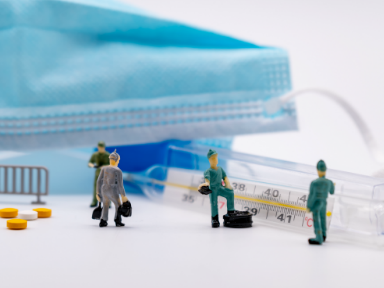
Global pharmaceutical company Mylan N.V. has announced the U.S. launch of Tadalafil Tablets USP, 20 mg, the first generic version of the reference listed drug, Eli Lilly and Company's Adcirca®.
Mylan Pharmaceuticals received final approval from the U.S. Food and Drug Administration (FDA) for its Abbreviated New Drug Application (ANDA) and was awarded 180 days of marketing exclusivity for this product, which is indicated for the treatment of pulmonary arterial hypertension to improve exercise ability.
U.S. sales for Tadalafil Tablets USP, 20 mg, were approximately $510 million for the 12 months ending May 31, 2018, according to IQVIA.
Currently, Mylan has 192 ANDAs pending FDA approval representing approximately $88.3 billion in annual brand sales, according to IQVIA. Forty-four of these pending ANDAs are potential first-to-file opportunities, representing $44.9 billion in annual brand sales, for the 12 months ending December 31, 2017, according to IQVIA.
-----------------------------------------------------------------------------
Editor's Note:
To apply for becoming a contributor of en-CPhI.cn,
welcome to send your CV and sample works to us,
Email: Julia.Zhang@ubmsinoexpo.com.




 ALL
ALL Pharma in China
Pharma in China Pharma Experts
Pharma Experts Market News
Market News Products Guide
Products Guide Brand Story
Brand Story































 Pharma Sources Insight January 2025
Pharma Sources Insight January 2025








